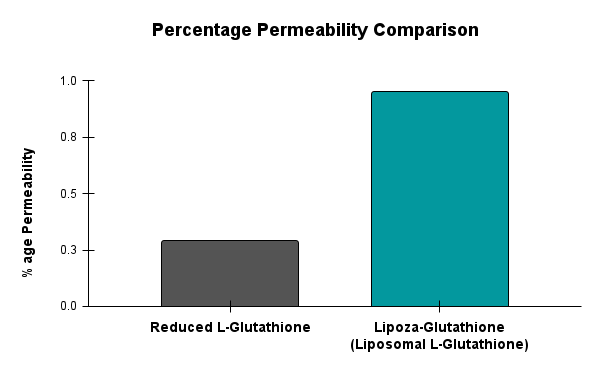
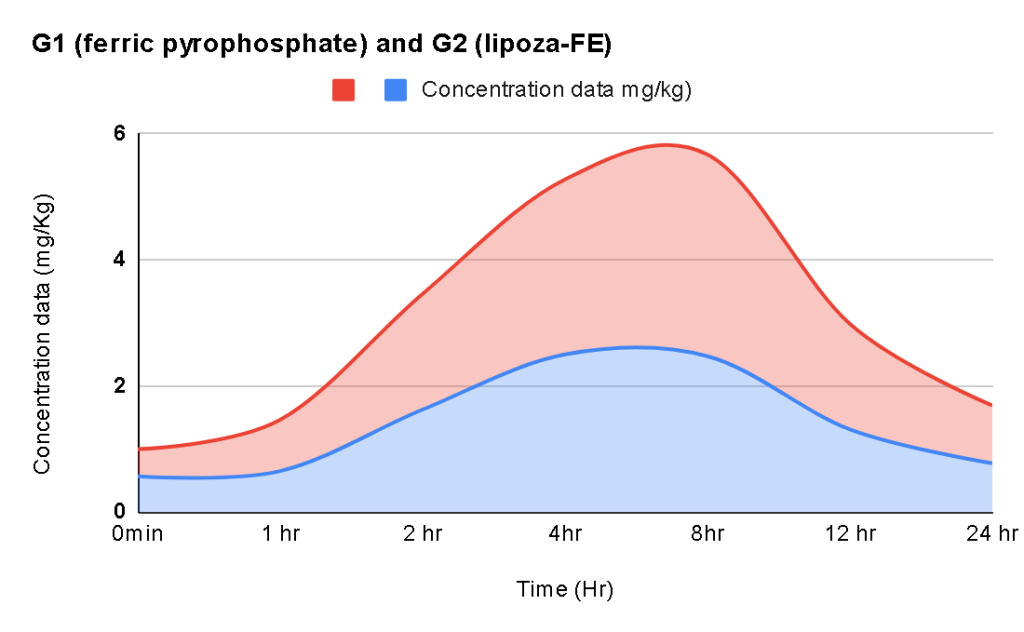
Upcoming Events
Vitafoods Europe 2026
We are presenting our latest innovations and look forward to connecting with industry professionals at the upcoming event.
- Fira Barcelona Gran Via, Barcelona, Spain
- 5 – 7 May 2026
- 10:00 AM
- Fira Barcelona Gran Via, Barcelona, Spain
- 5 – 7 May 2026
- 10:00 AM
Connect with our team and explore new opportunities.
Previous Events
Take a look at the events and exhibitions where Ozabio has previously participated and connected with industry professionals.

OZABIO at Vitafoods India 2026 was a successful showcase of innovation and nutraceutical solutions. The event provided a great opportunity to connect with industry professionals and present our latest liposomal technologies.

OZABIO at Vitafoods India 2026: A Breakthrough Showcase of Innovation & Impact OZABIO delivered an outstanding presence at Vitafoods India 2026, marking one of our most successful exhibitions to date.

OZABIO at Vitafoods Asia 2025: Showcasing the Future of Nutraceutical Innovation OZABIO proudly participated in Vitafoods Asia 2025, one of the region’s most influential platforms for nutraceuticals and functional ingredients.

OZABIO at Vitafoods Europe 2025: Elevating Nutraceutical Performance on a Global Stage OZABIO marked a powerful presence at Vitafoods Europe 2025, the world’s leading exhibition for nutraceutical innovation.
Connect With Our Team
Have questions about our products or services? Reach out to us and our team will be happy to assist you with the right information and support.
- OzaBio is a global provider of high-quality nutritional ingredients, specializing in advanced liposomal and functional nutraceutical solutions. We are committed to innovation, quality, and improved bioavailability to support modern health and wellness needs.
Phone: +91 78769 66742, +1 (571)315-3939, +971 52 156 2603
Mail: [email protected]
Location: 1207 Delaware Ave #2877, Wilmington DE 19806, USA
© 2026 OzaBio | All Rights Reserved.