- Barcelona, Spain
- 20–22 May 2025
- Stall No. 5J23
OZABIO at Vitafoods Europe 2025: Elevating Nutraceutical Performance on a Global Stage
OZABIO marked a powerful presence at Vitafoods Europe 2025, the world’s leading exhibition for nutraceutical innovation. As a global meeting point for scientific advancement, ingredient innovation, and market-leading brands, the event provided an exceptional platform to showcase the future of high-performance nutrition.
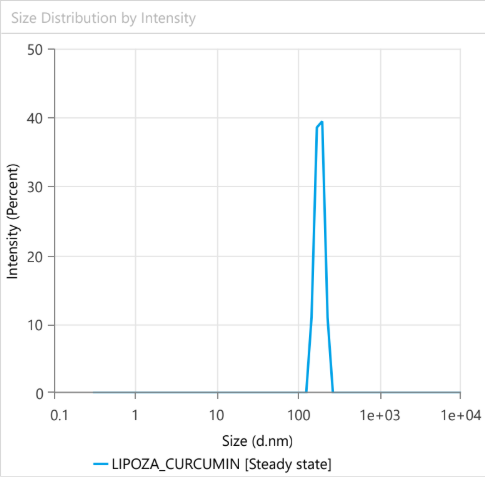
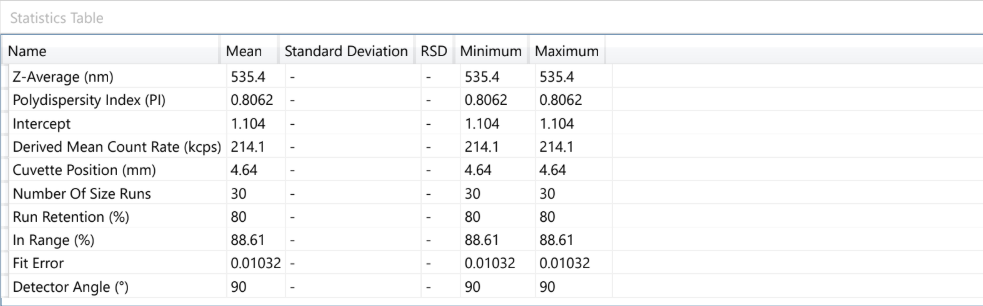
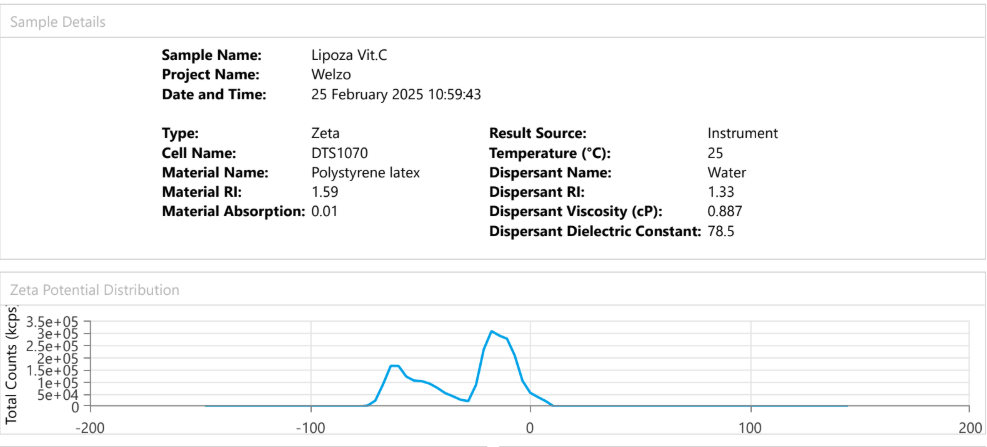
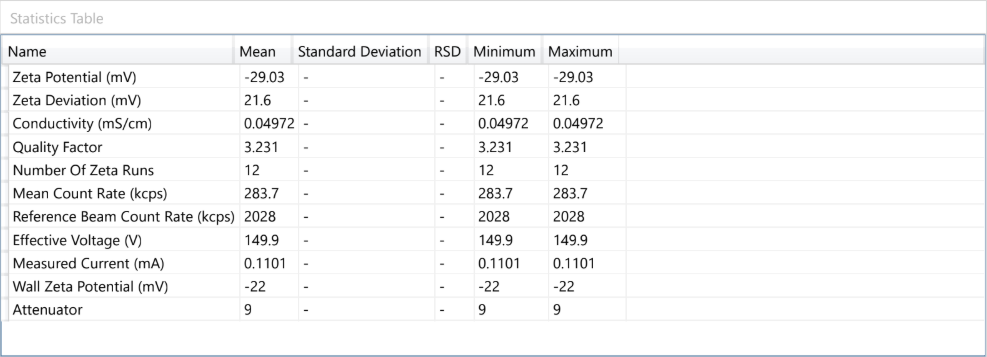
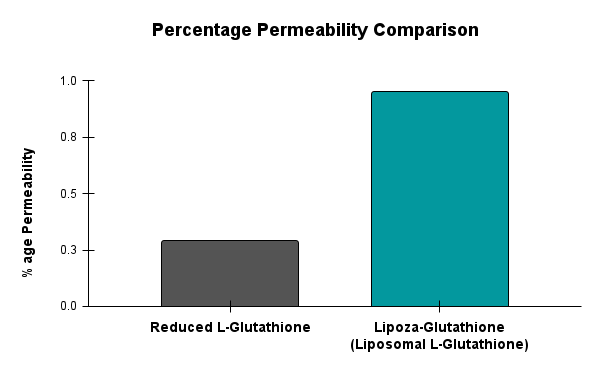
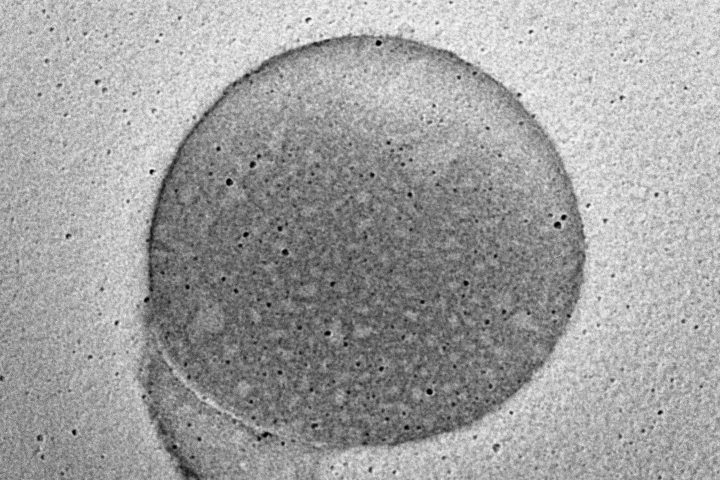
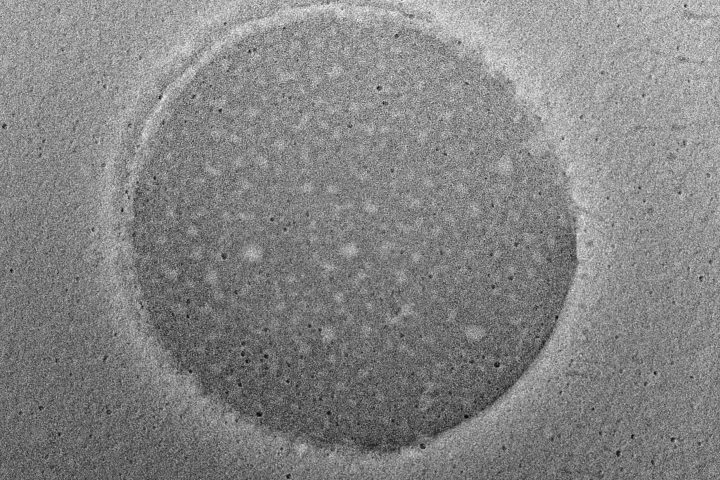
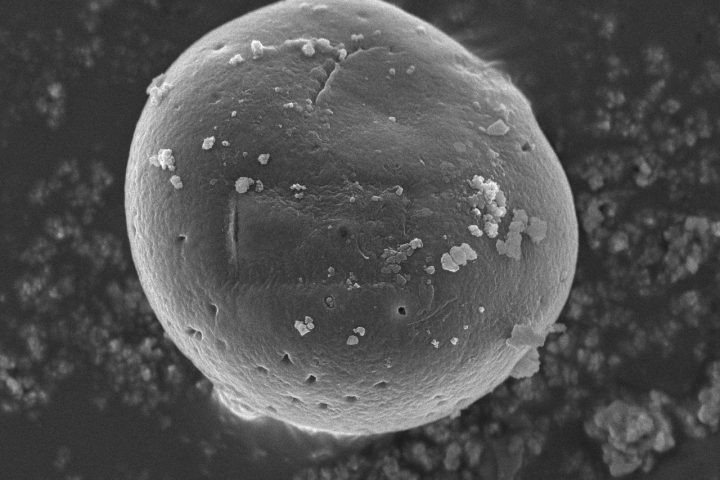
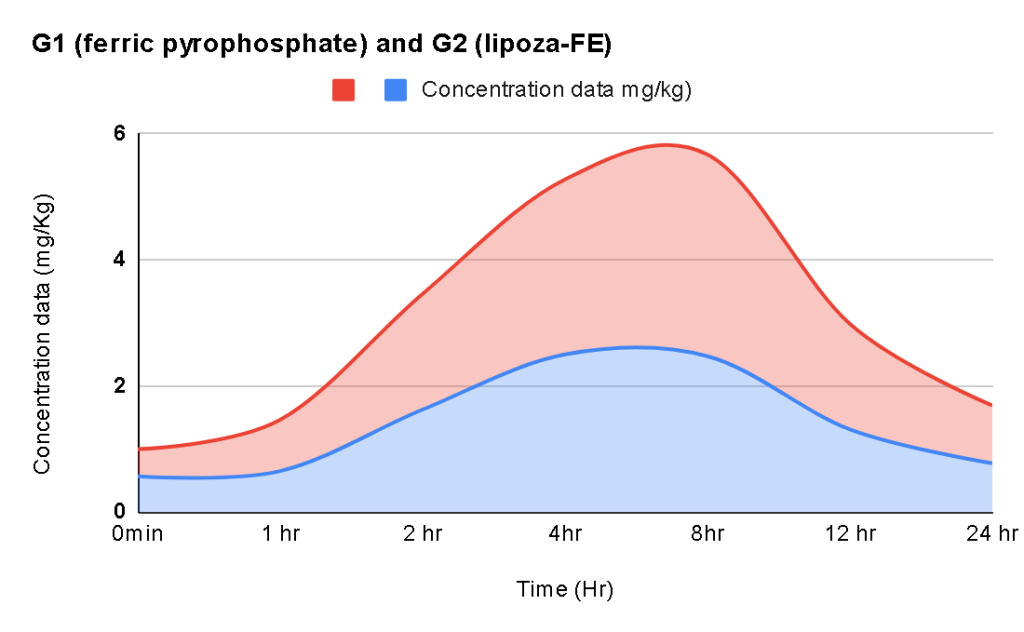
At the forefront of our presentation was LIPOZA, OZABIO’s advanced liposomal delivery platform—engineered to redefine how nutrients are absorbed, protected, and delivered. Our solutions attracted strong interest from international partners seeking next-generation formulations with superior bioavailability, enhanced stability, and measurable efficacy.
Global Engagement & Strategic Impact
Vitafoods Europe 2025 enabled OZABIO to:
● Build connections with key global stakeholders and decision-makers
● Showcase cutting-edge liposomal innovations to a highly specialized audience
● Explore collaborations across Europe, North America, and emerging markets
● Engage in high-level discussions on future trends, regulatory evolution, and consumer expectations
Our participation reinforced OZABIO’s position as a forward-thinking, innovation-driven partner for brands aiming to lead in a competitive global landscape.
Beyond Participation: Setting New Benchmarks
More than just an exhibition, Vitafoods Europe was a platform for OZABIO to demonstrate its commitment to scientific excellence and continuous innovation. By aligning our technology with global market needs, we continue to empower brands to create differentiated, high-impact nutraceutical products.
- OzaBio is a global provider of high-quality nutritional ingredients, specializing in advanced liposomal and functional nutraceutical solutions. We are committed to innovation, quality, and improved bioavailability to support modern health and wellness needs.
Phone: +91 78769 66742, +1 (571)315-3939, +971 52 156 2603
Mail: [email protected]
Location: 1207 Delaware Ave #2877, Wilmington DE 19806, USA
© 2026 OzaBio | All Rights Reserved.