- Bangkok, Thailand
- 17–19 Sept 2025
- Stall No. A63
OZABIO at Vitafoods Asia 2025: Showcasing the Future of Nutraceutical Innovation
OZABIO proudly participated in Vitafoods Asia 2025, one of the region’s most influential platforms for nutraceuticals and functional ingredients. The event brought together industry leaders, innovators, and forward-thinking brands, creating a dynamic space to explore the future of health and nutrition.
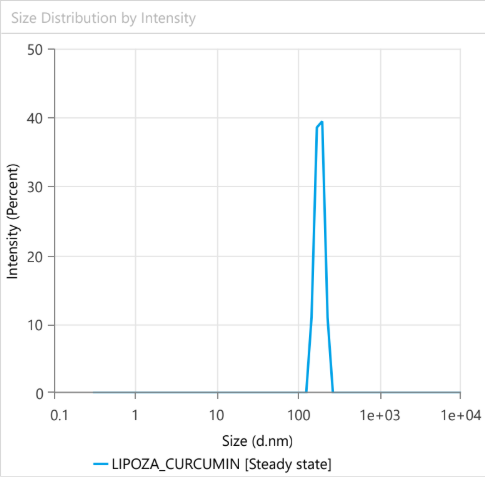
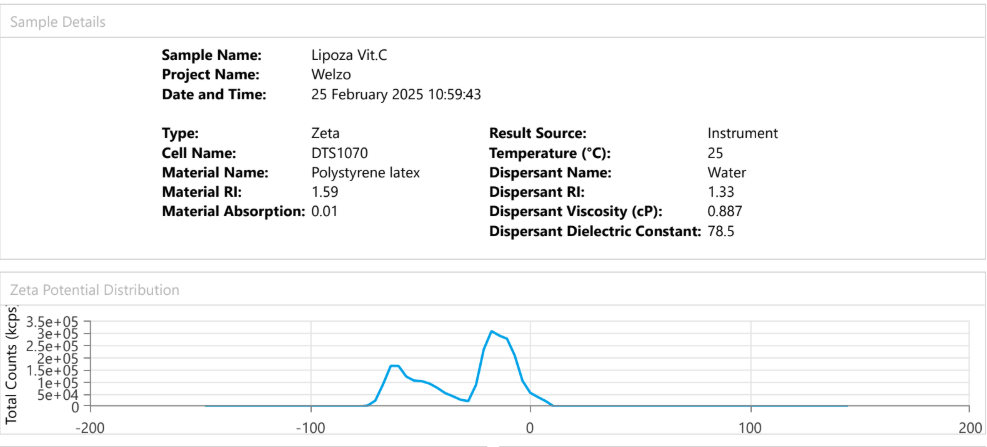
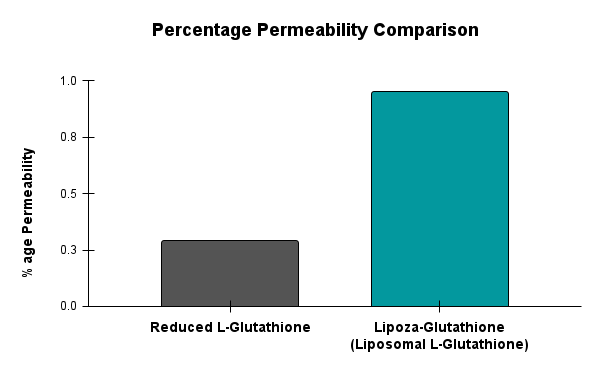
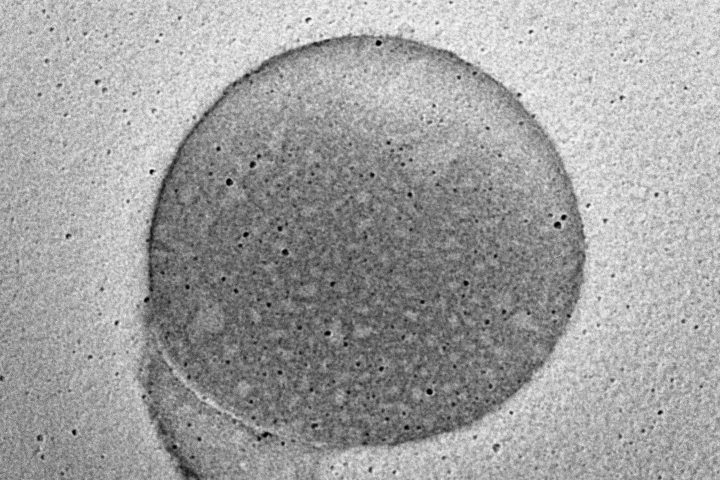
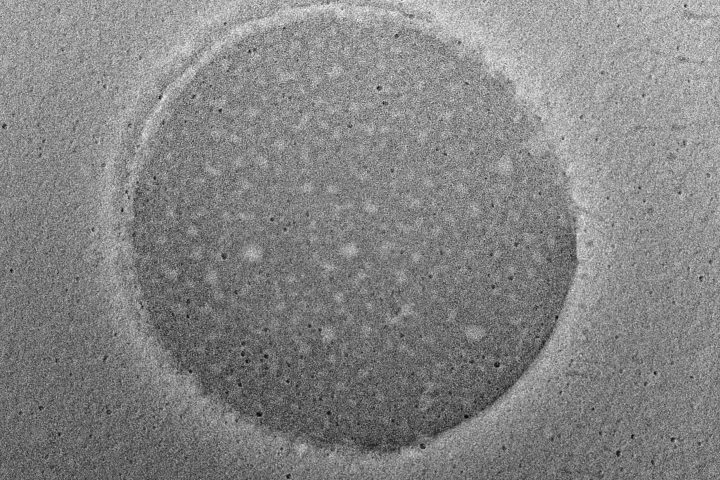
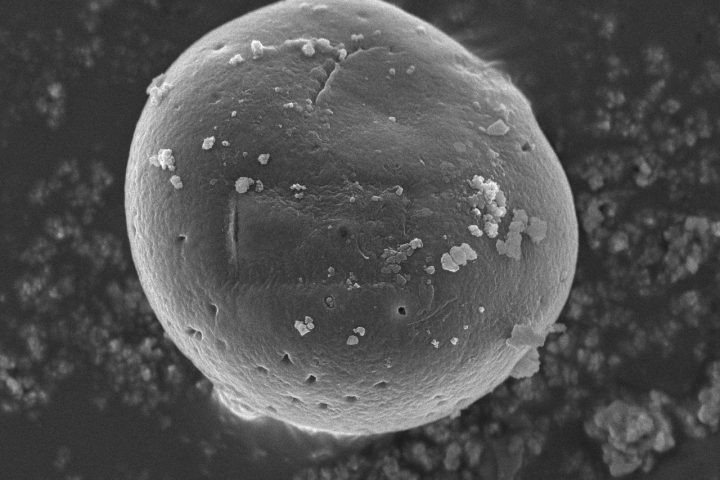
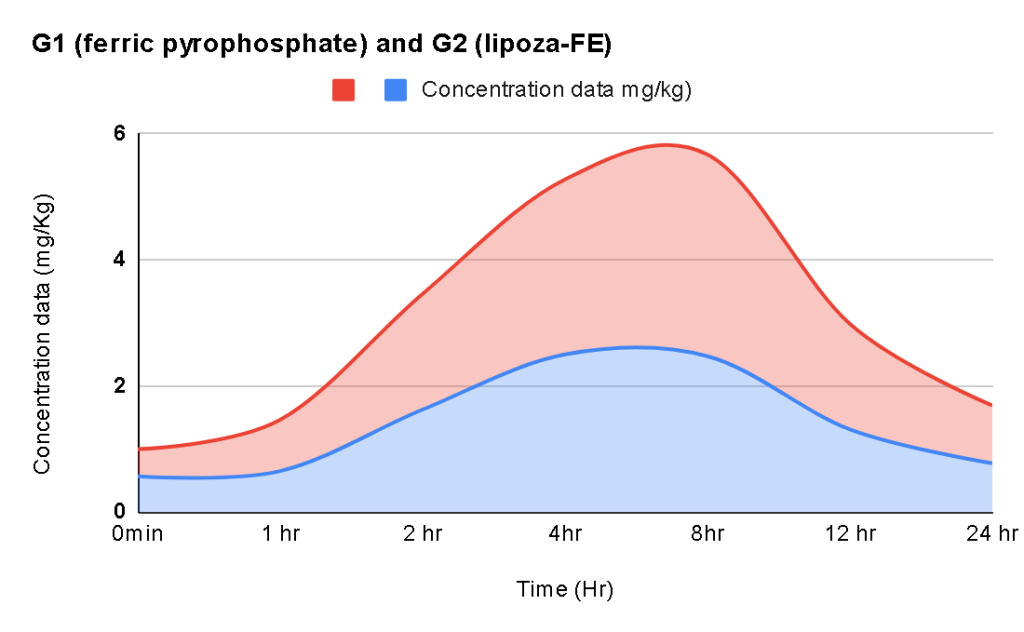
At the exhibition, OZABIO showcased its advanced LIPOZA liposomal technology, highlighting how next-generation delivery systems are transforming ingredient performance through enhanced bioavailability, stability, and targeted nutrient delivery. Our presence reflected our commitment to driving science-backed innovation and delivering solutions that go beyond conventional formulations.
Our Experience & Key Highlights
Participating in Vitafoods Asia 2025 provided OZABIO with a valuable opportunity to:
● Connect with global partners, manufacturers, and nutraceutical brands
● Present our cutting-edge liposomal solutions to a wider audience
● Engage in meaningful discussions around emerging trends, regulatory developments, and market demands
The event reinforced OZABIO’s position as a trusted innovation partner, focused on enabling brands to create high-performance, differentiated products.
Driving Innovation Beyond the Event
Our participation at Vitafoods Asia reflects OZABIO’s ongoing mission to advance nutraceutical science and strengthen global collaborations. Events like these allow us to stay at the forefront of industry evolution while continuously refining our technologies to meet the changing needs of the market.
- OzaBio is a global provider of high-quality nutritional ingredients, specializing in advanced liposomal and functional nutraceutical solutions. We are committed to innovation, quality, and improved bioavailability to support modern health and wellness needs.
Phone: +91 78769 66742, +1 (571)315-3939, +971 52 156 2603
Mail: [email protected]
Location: 1207 Delaware Ave #2877, Wilmington DE 19806, USA
© 2026 OzaBio | All Rights Reserved.