Comprehensive Evaluation of Liposomal Formulations
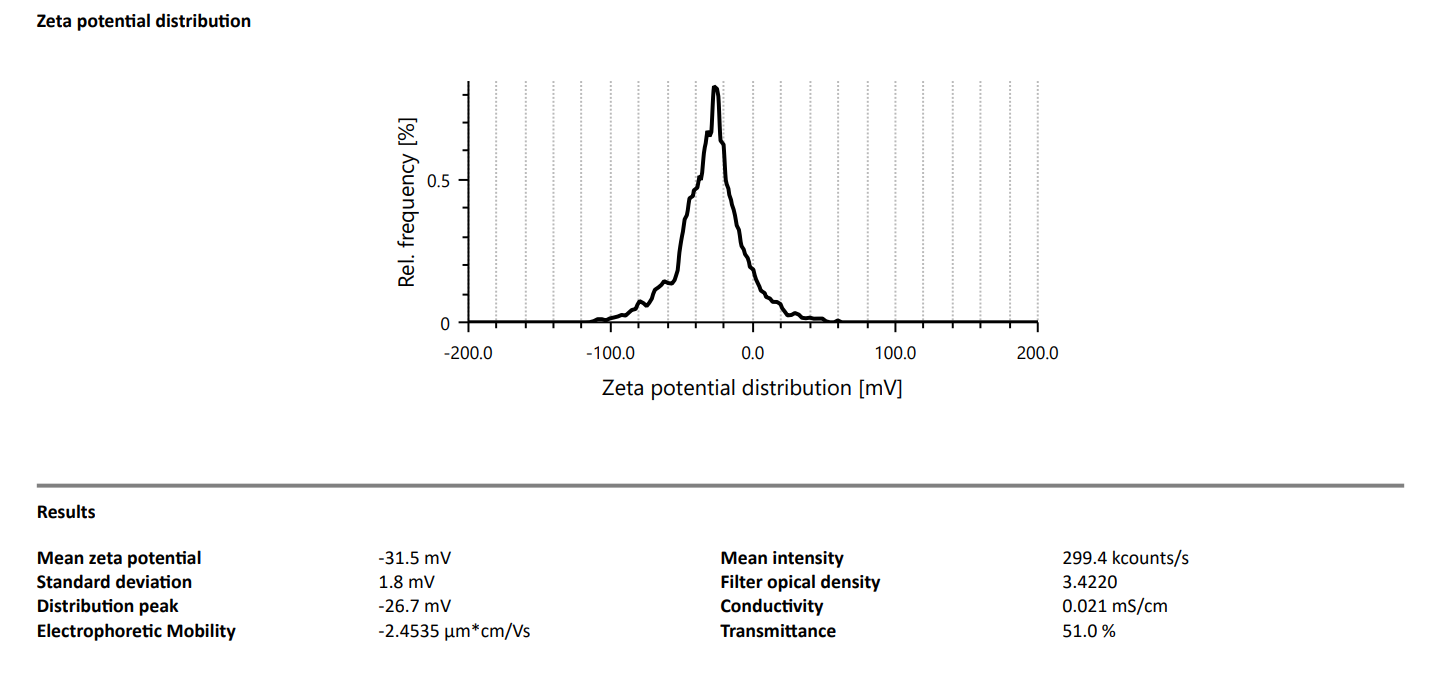
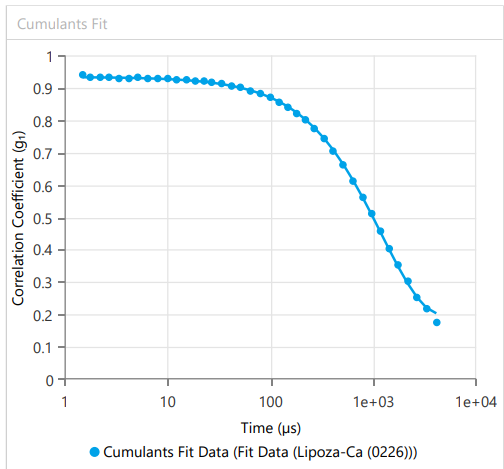
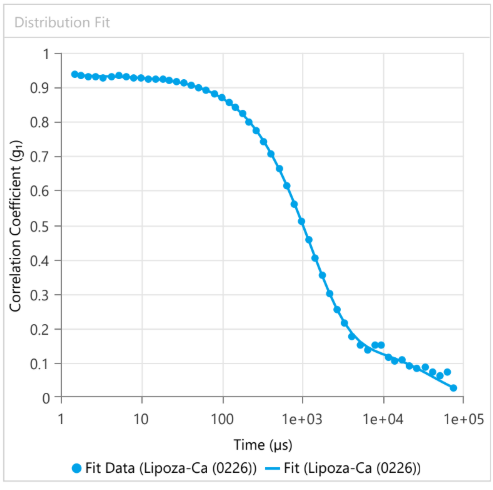
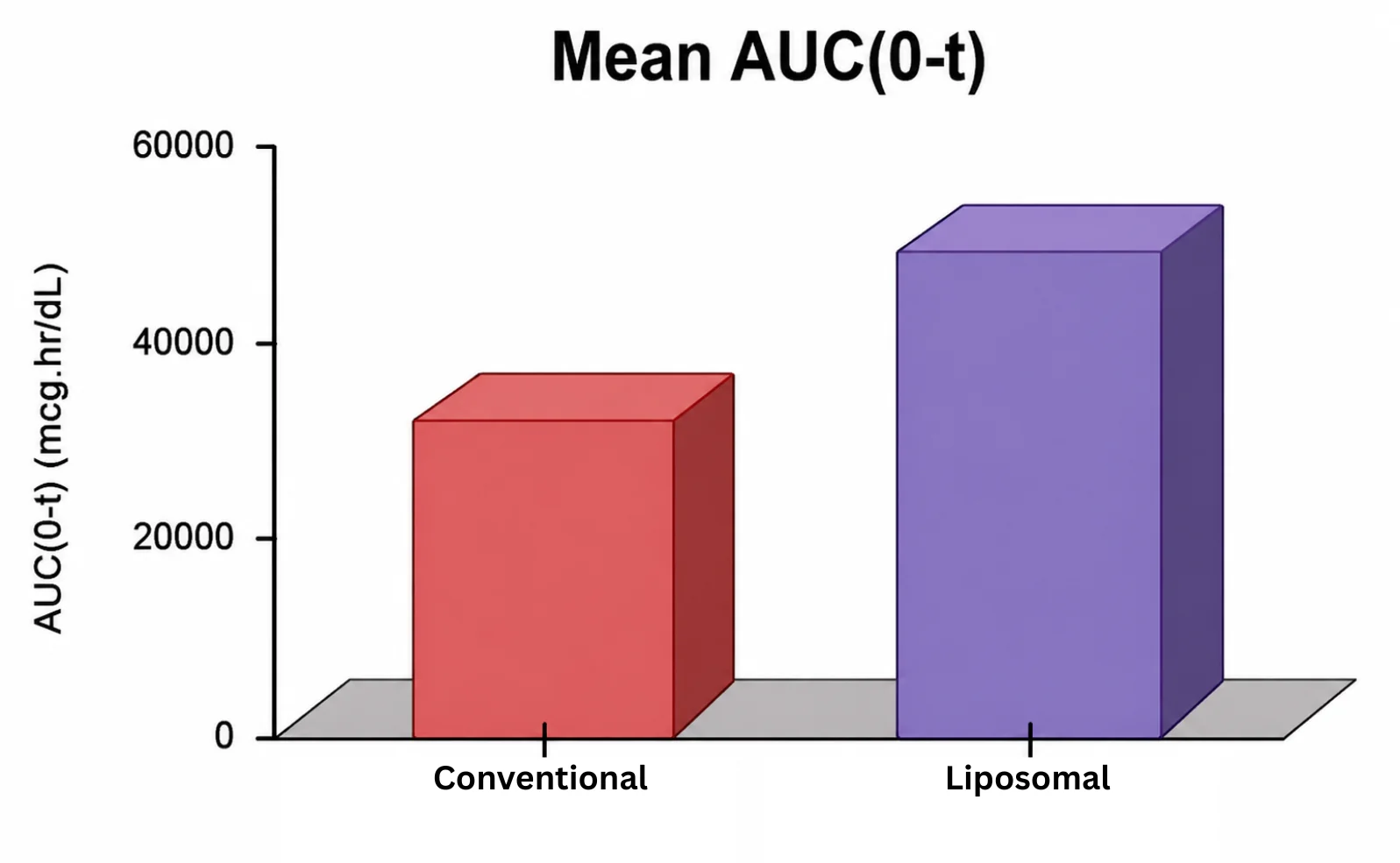
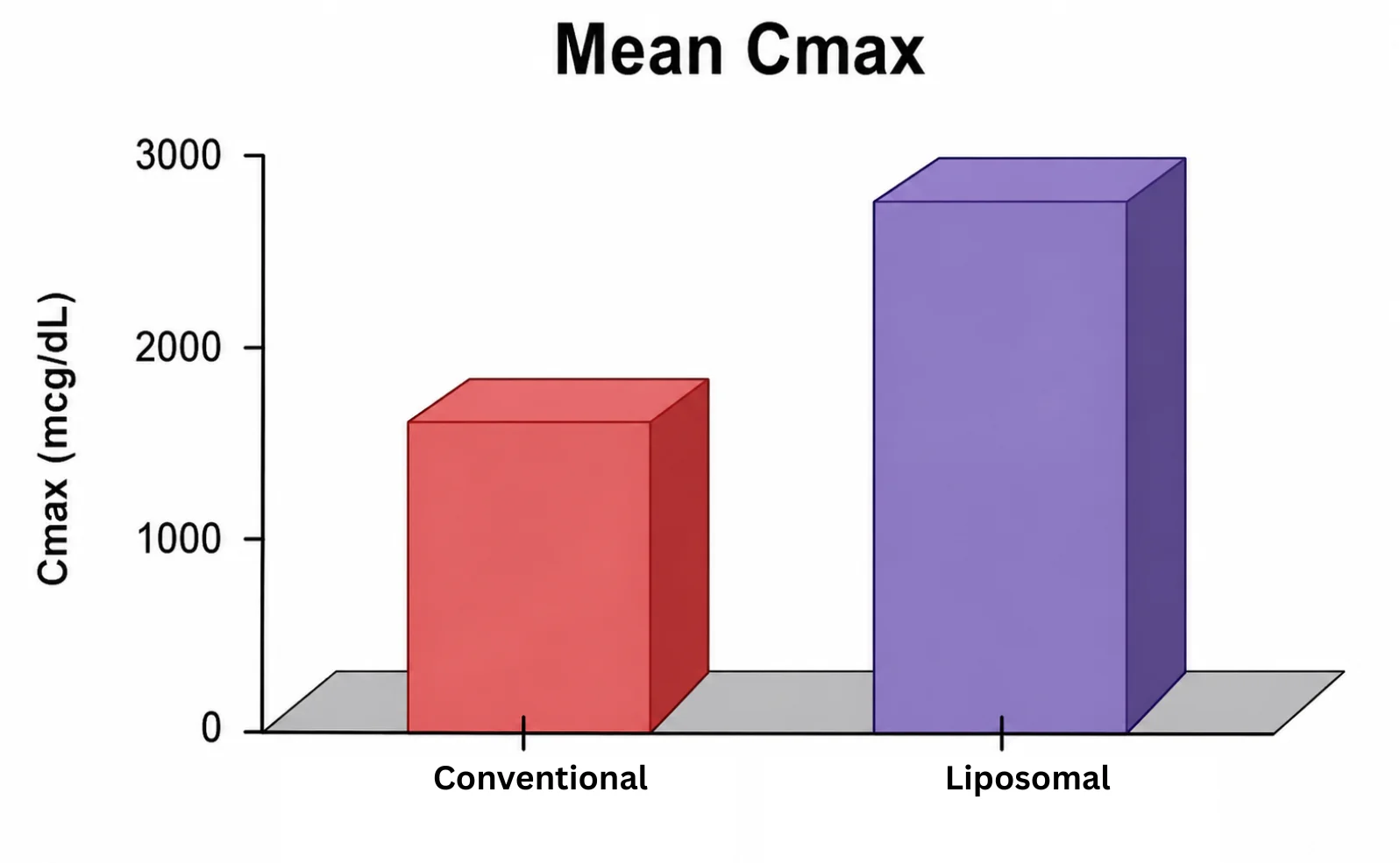
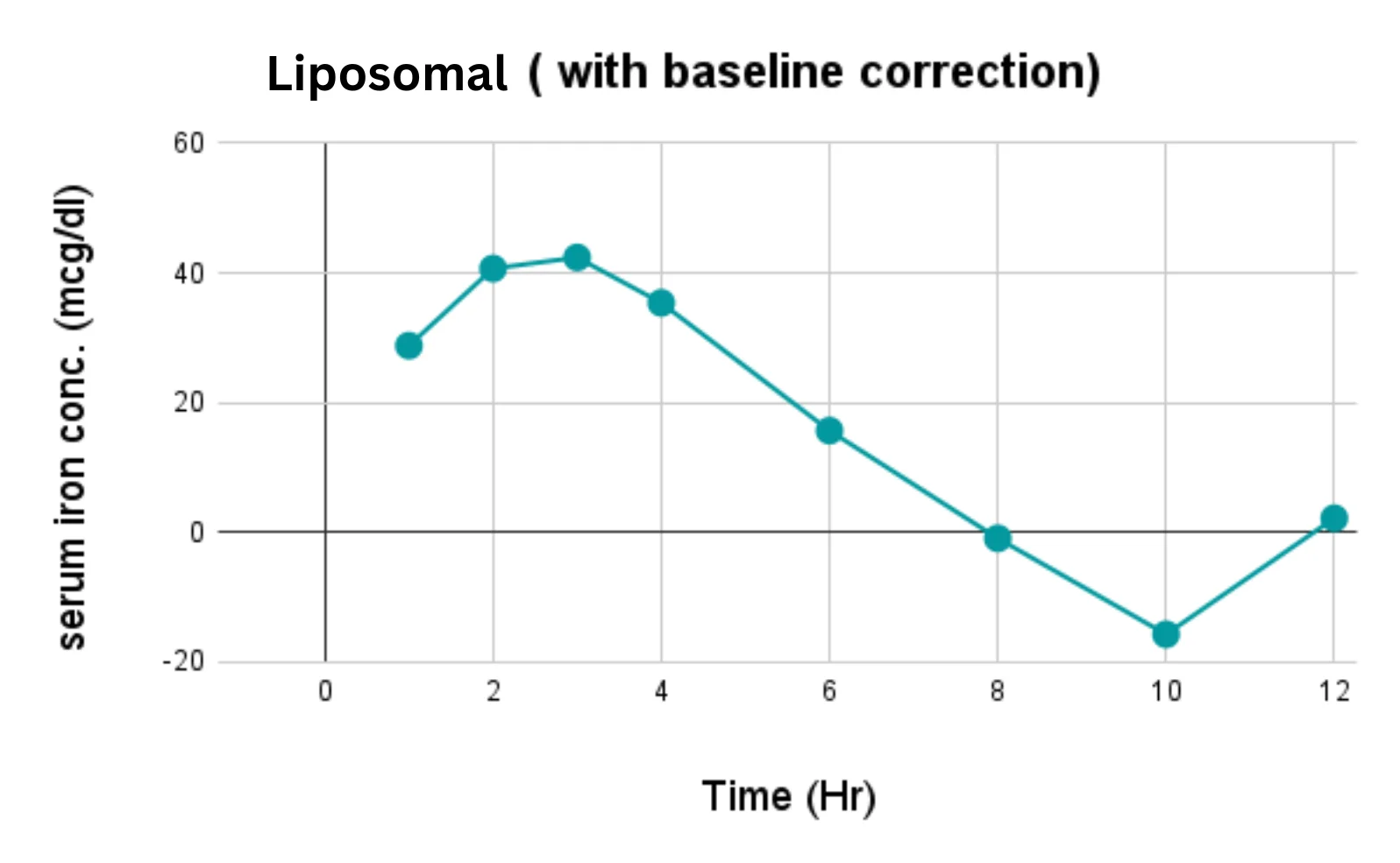
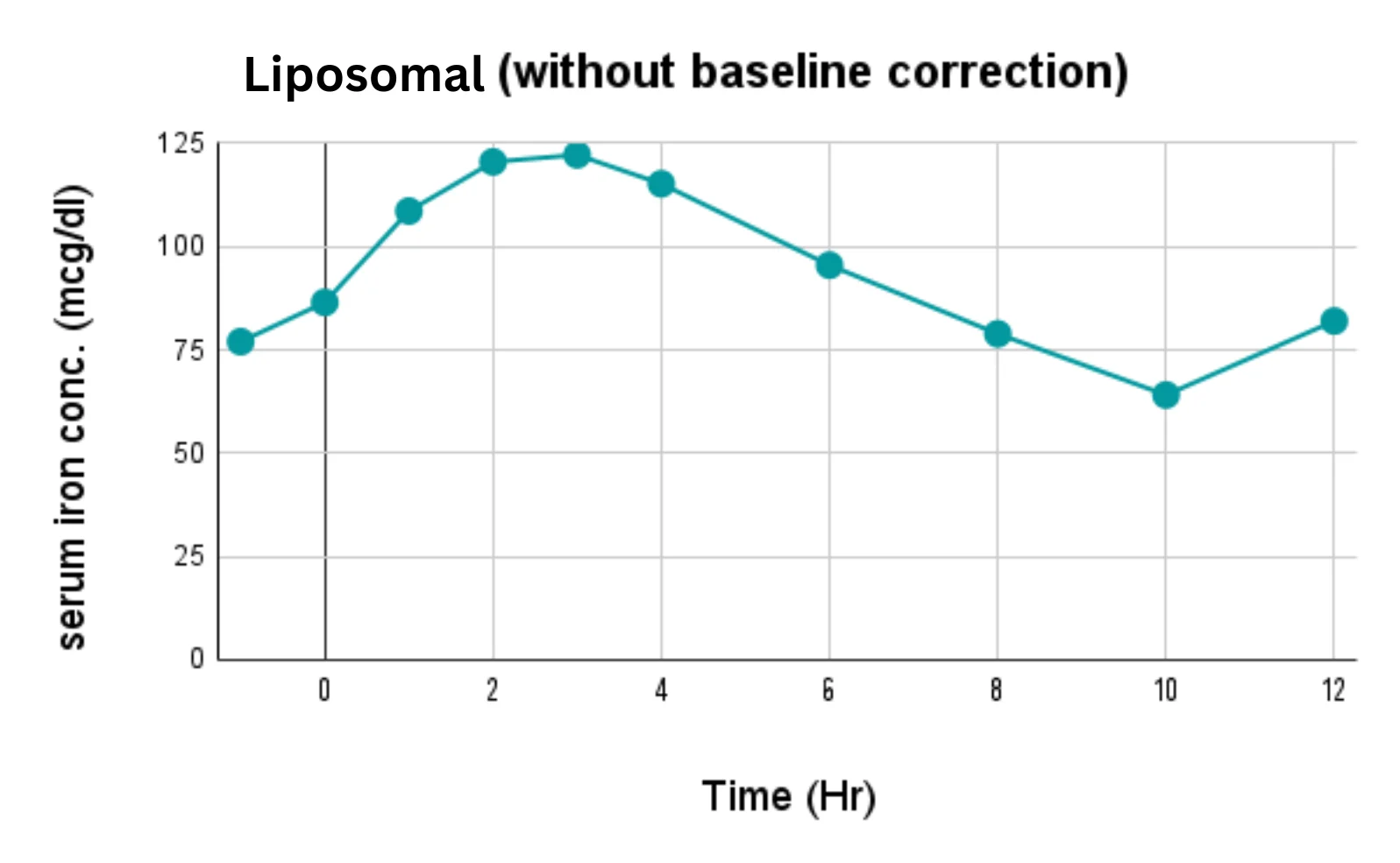
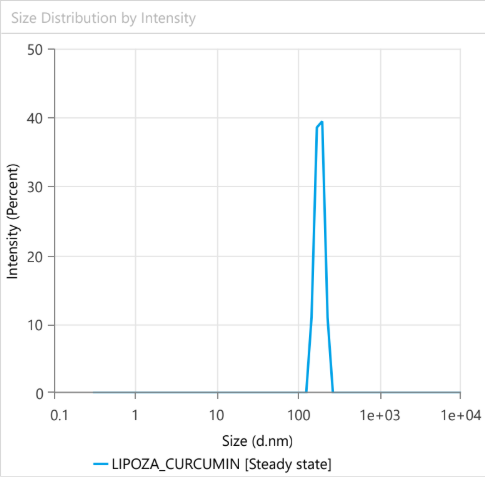
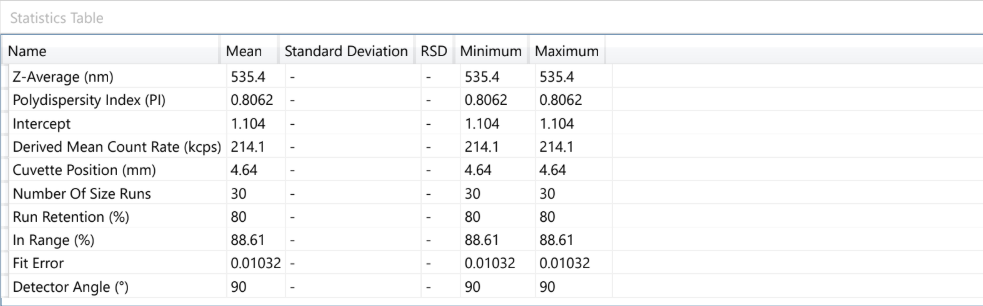
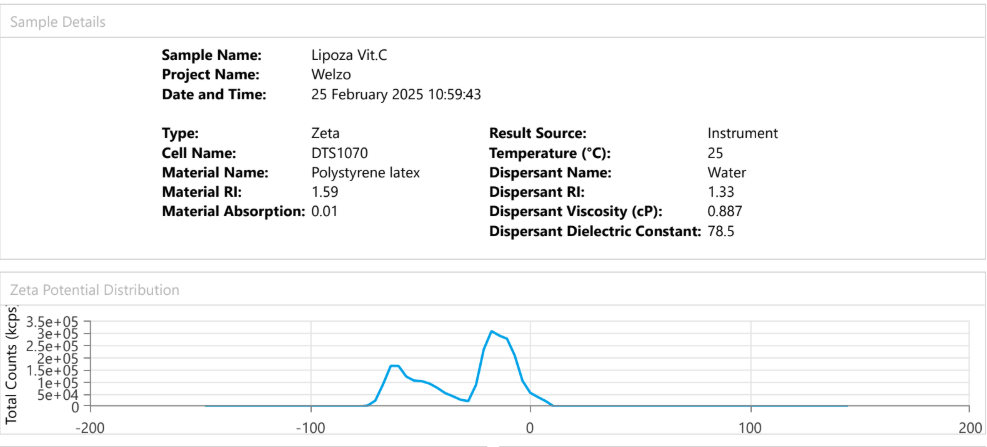
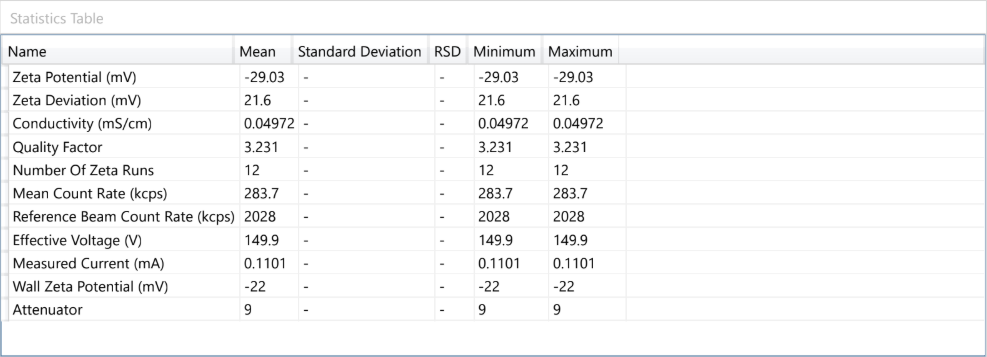
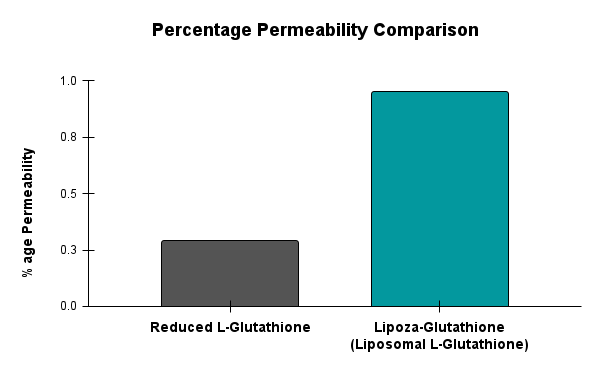
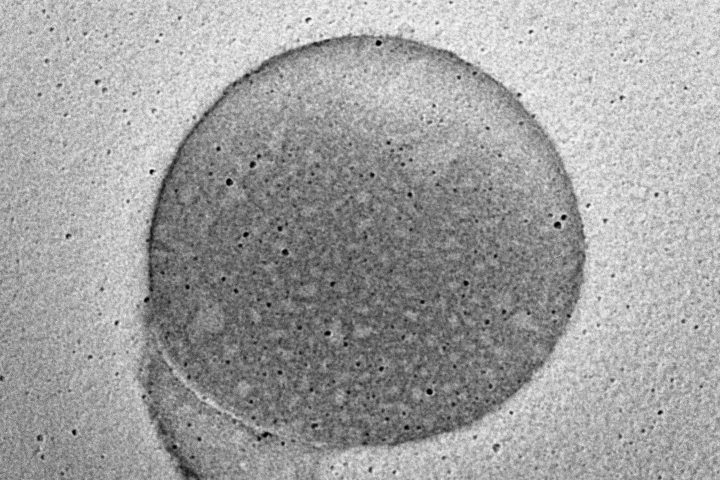
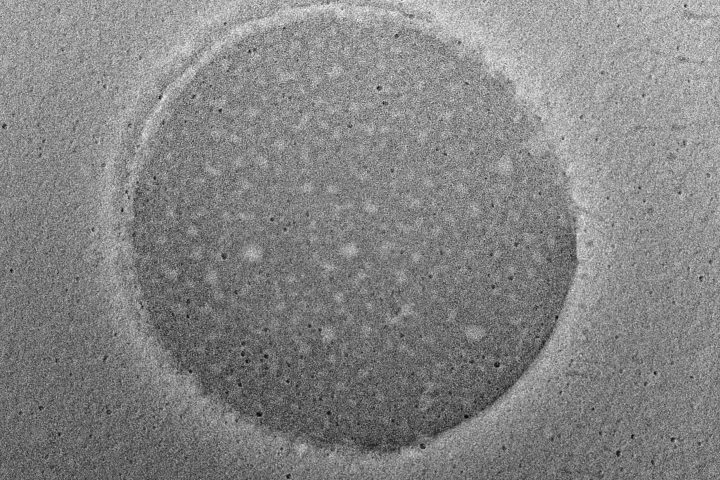
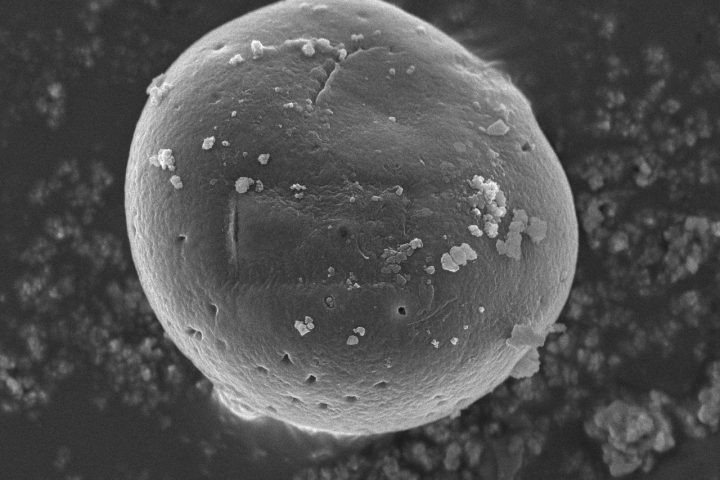
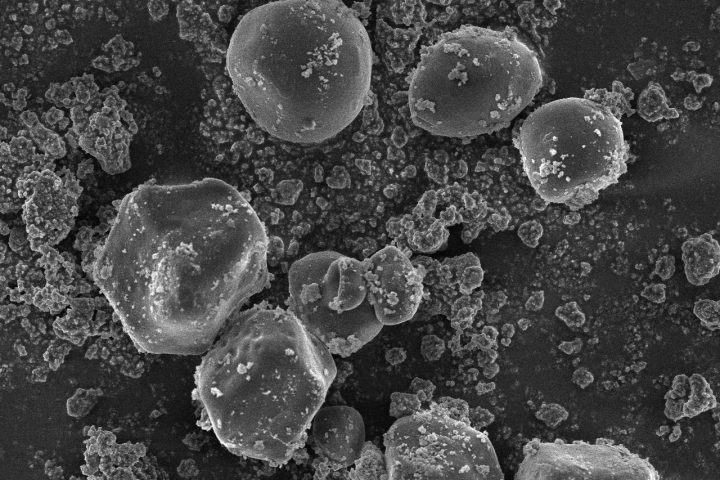
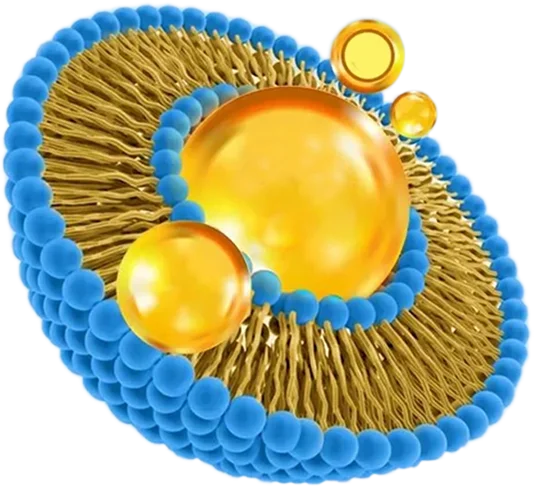
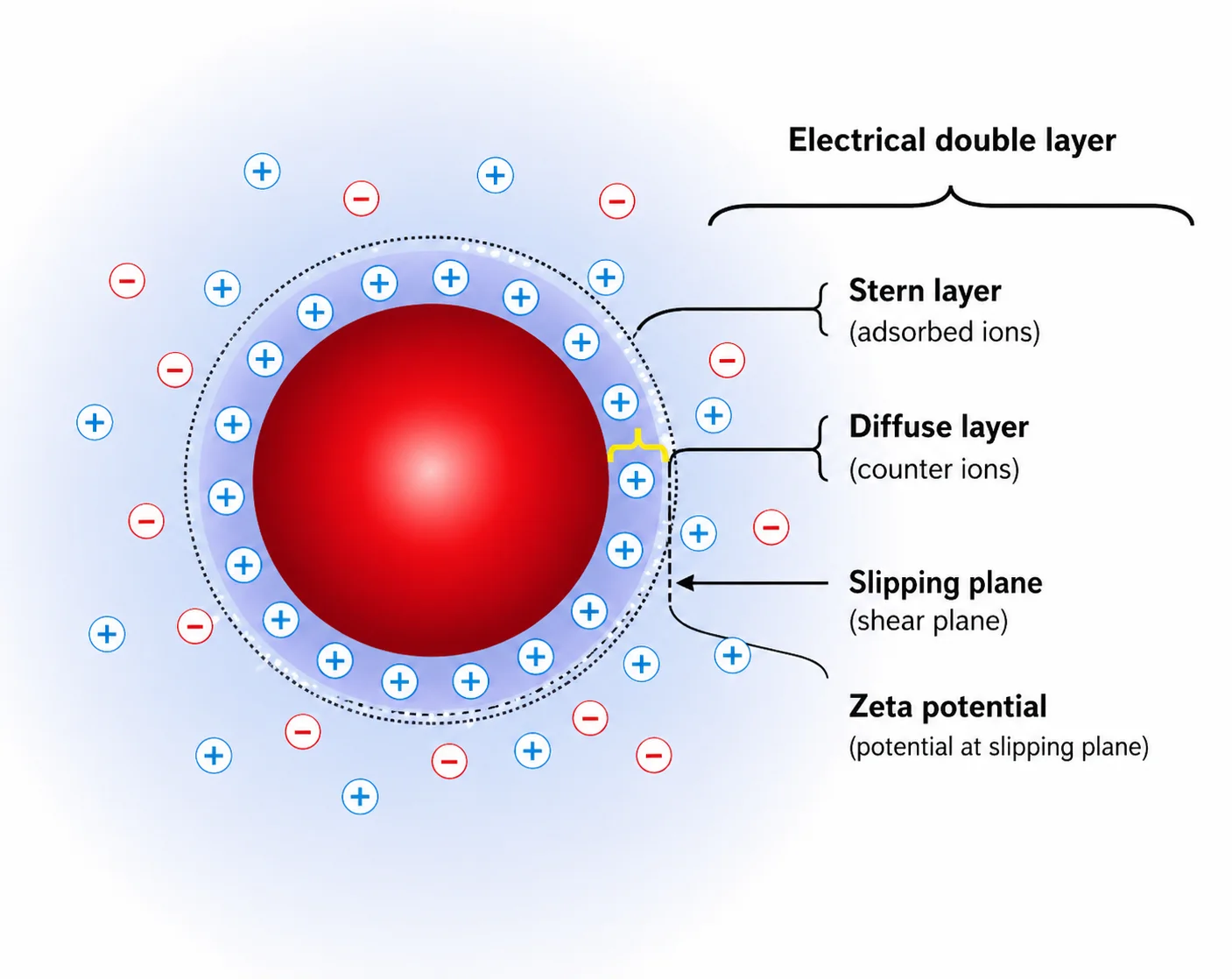
At OzaBio, our Research & Development is driven by a rigorous, science-led approach focused on developing high-performance liposomal nutraceutical solutions with enhanced bioavailability and functional efficiency. Each formulation undergoes a comprehensive and systematic evaluation, beginning with detailed physicochemical characterization, including particle size distribution, zeta potential, and encapsulation efficiency, which are critical to ensuring stability and effective delivery. This is complemented by in-vitro performance assessments such as dissolution profiling and permeability studies, along with comparative analysis against conventional delivery systems to validate improved absorption dynamics. Our formulations are further designed to exhibit controlled and sustained release behavior, ensuring optimal nutrient protection and delivery under physiological conditions. Extensive stability and compatibility studies are conducted under varied environmental parameters to confirm product integrity, robustness, and shelf-life reliability. Where applicable, formulations are supported by preclinical and human-based evaluations to strengthen safety and performance validation. Backed by advanced analytical techniques and aligned with globally accepted quality standards, our R&D framework ensures consistency, reliability, and innovation, enabling us to deliver scientifically validated liposomal solutions tailored for diverse nutraceutical applications.
Comprehensive Evaluation of Liposomal Formulations
At OzaBio, our Research & Development is driven by a rigorous, science-led approach focused on developing high-performance liposomal nutraceutical solutions with enhanced bioavailability and functional efficiency. Each formulation undergoes a comprehensive and systematic evaluation, beginning with detailed physicochemical characterization, including particle size distribution, zeta potential, and encapsulation efficiency, which are critical to ensuring stability and effective delivery. This is complemented by in-vitro performance assessments such as dissolution profiling and permeability studies, along with comparative analysis against conventional delivery systems to validate improved absorption dynamics. Our formulations are further designed to exhibit controlled and sustained release behavior, ensuring optimal nutrient protection and delivery under physiological conditions. Extensive stability and compatibility studies are conducted under varied environmental parameters to confirm product integrity, robustness, and shelf-life reliability. Where applicable, formulations are supported by preclinical and human-based evaluations to strengthen safety and performance validation. Backed by advanced analytical techniques and aligned with globally accepted quality standards, our R&D framework ensures consistency, reliability, and innovation, enabling us to deliver scientifically validated liposomal solutions tailored for diverse nutraceutical applications.
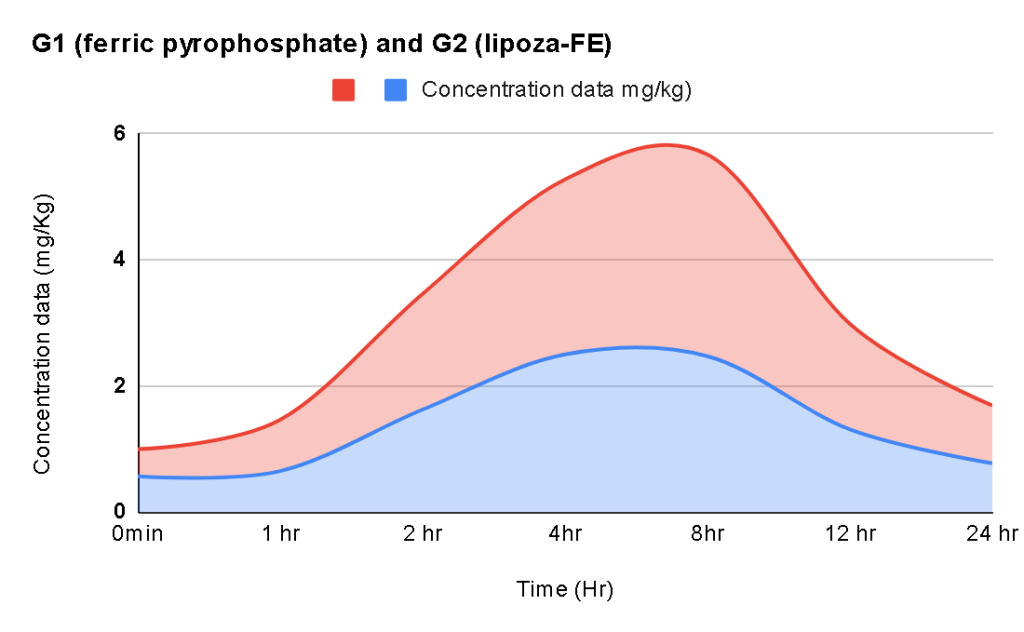
Research & Studies
Our formulations are supported by detailed scientific studies, including:



- OzaBio is a global provider of high-quality nutritional ingredients, specializing in advanced liposomal and functional nutraceutical solutions. We are committed to innovation, quality, and improved bioavailability to support modern health and wellness needs.
Phone: +91 78769 66742, +1 (571)315-3939, +971 52 156 2603
Mail: [email protected]
Location: 1207 Delaware Ave #2877, Wilmington DE 19806, USA
© 2026 OzaBio | All Rights Reserved.