FAQs
Frequently Asked Questions
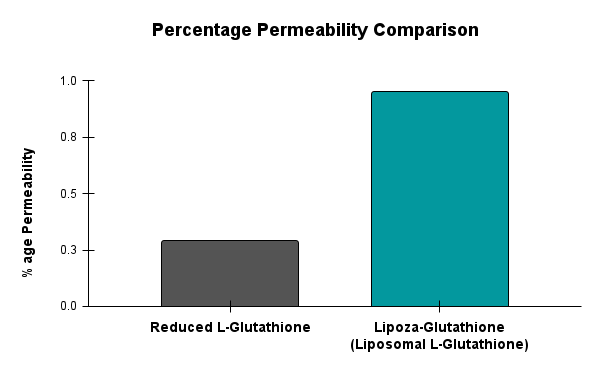
OZABIO specializes in next-generation nutraceutical innovation, delivering advanced liposomal ingredients that redefine how nutrients are absorbed and utilized. With its LIPOZA technology, OZABIO enables brands to create high-impact, highly bioavailable formulations that outperform conventional products helping you stand out in a competitive market.
Unlike conventional forms that often degrade in the digestive system, LIPOZA ingredients are designed for maximum bioavailability. This means:
- Higher absorption
- Faster action
- Improved efficacy at lower doses
Yes. Liposomes are typically made from phospholipids, which are naturally found in the human body. When formulated correctly (like in LIPOZA products), they are safe, biocompatible, and well-tolerated.
LIPOZA ingredients are designed for a wide range of industries and product innovators looking to create high-performance formulations. They are ideal for:
- Nutraceutical brands – to develop supplements with superior absorption and efficacy
- Pharmaceutical companies – for advanced drug delivery and improved therapeutic outcomes
- Functional food & beverage manufacturers – to create next-gen fortified products with better bioavailability
- Sports nutrition brands – for faster nutrient uptake and performance-focused formulations
- Cosmeceutical & beauty brands – for skin, hair, and anti-aging solutions with enhanced delivery
- Contract manufacturers & formulators – seeking innovative ingredients to offer differentiated products
- Wellness startups & D2C brands – looking to stand out with science-backed, premium formulations
- Whether you’re developing capsules, powders, liquids, gummies or functional beverages, LIPOZA enables you to upgrade your product’s effectiveness and market positioning.
Yes, OZABIO operates as a global nutraceutical ingredient supplier, serving partners across multiple countries and regions.
We support international clients with efficient logistics, documentation, and export handling, ensuring smooth delivery of LIPOZA ingredients worldwide. Shipping timelines and regulatory requirements may vary depending on the destination country and product specifications.
- For specific shipping details, our team will guide you through:
- Available shipping options
- Documentation & compliance requirements
- Estimated delivery timelines
- Simply reach out to us, and we’ll assist you with end-to-end international order support.
You can request a quotation by contacting us via website, email, or sales team. Share your requirements, and we’ll provide a customized quote with pricing and details quickly.
Liposomal ingredients can be used in:
Capsules & sachets
Liquid formulations
Functional beverages
Powders
Gummies
They are highly versatile and adaptable across multiple dosage formats.
Liposomal encapsulation often improves taste masking and enhances stability. This makes it easier to formulate products that are both effective and consumer-friendly.
Getting started is simple. Reach out to us with your requirements, and our team will assist you with:
- Product selection
- Sample requests
- Custom solutions
Let’s build your next best-selling formulation together.
Our MOQ depends on the specific ingredient and application. We aim to be flexible and can accommodate both pilot-scale requirements and large commercial volumes, making it easier for brands to scale with us.
Yes, we provide samples to help you evaluate quality, compatibility, and performance before committing to bulk production. This ensures confidence in your final formulation.
Yes, we support urgent orders based on availability, with priority processing and faster shipping options to meet tight deadlines.
Yes, OZABIO is committed to maintaining high-quality and globally compliant ingredients. Our LIPOZA range is developed following strict quality control protocols and industry best practices, ensuring safety, consistency, and reliability.
We support compliance by providing:
- Complete documentation (COAs, technical data sheets)
- Quality and safety testing reports
- Guidance aligned with international regulatory requirements
This ensures our ingredients can be confidently used across multiple global markets.
- OzaBio is a global provider of high-quality nutritional ingredients, specializing in advanced liposomal and functional nutraceutical solutions. We are committed to innovation, quality, and improved bioavailability to support modern health and wellness needs.
Phone: +91 78769 66742, +1 (571)315-3939, +971 52 156 2603
Mail: [email protected]
Location: 1207 Delaware Ave #2877, Wilmington DE 19806, USA
© 2026 OzaBio | All Rights Reserved.