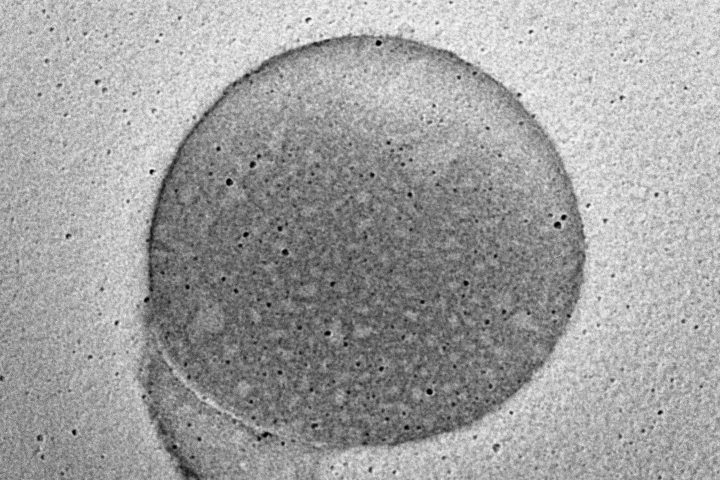
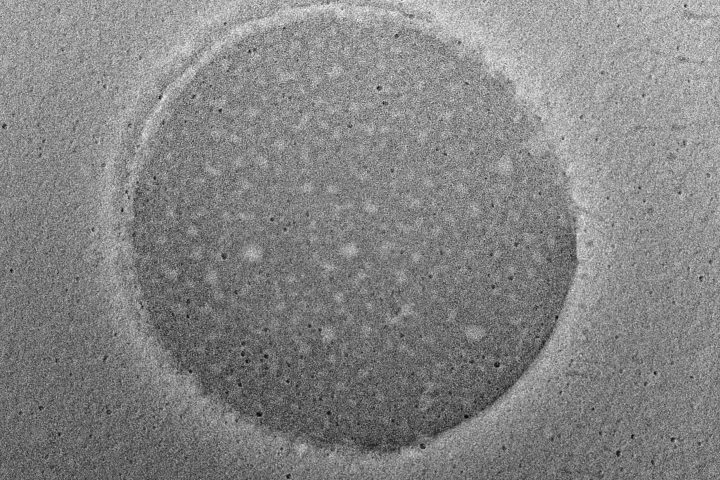
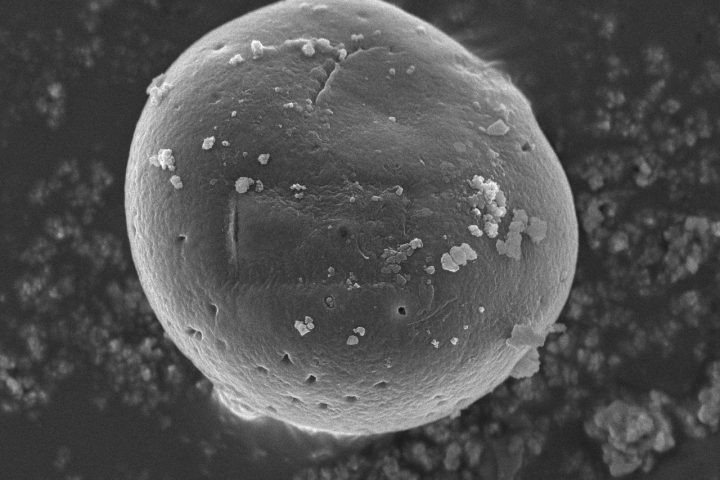
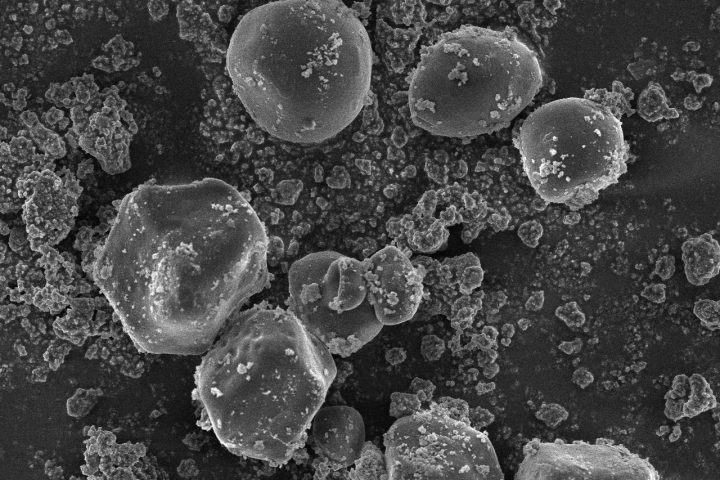
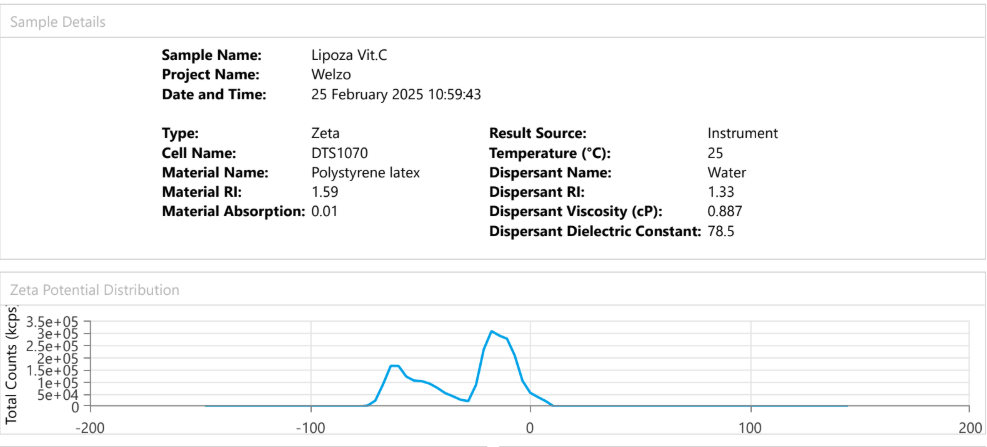
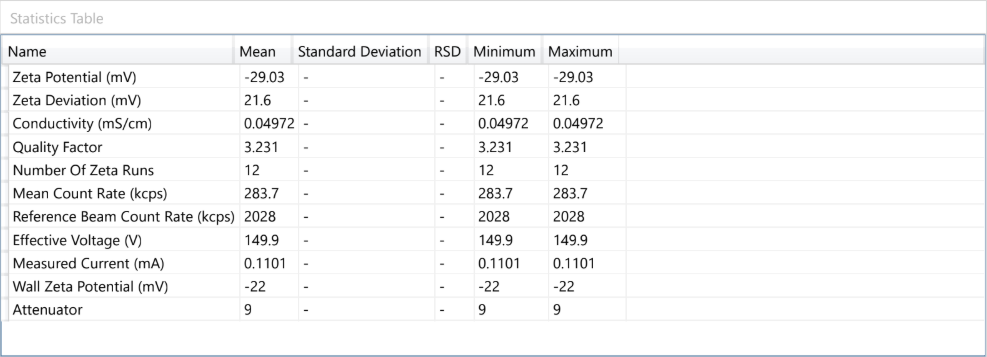
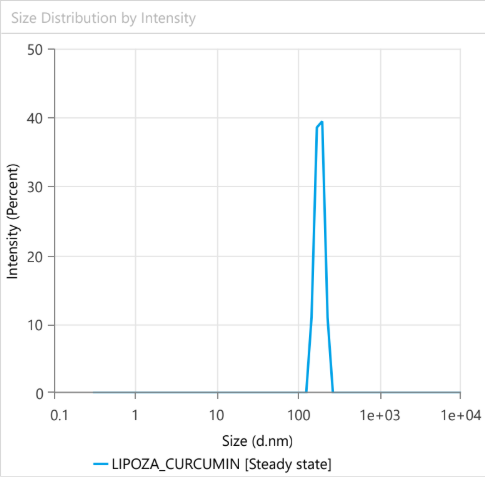
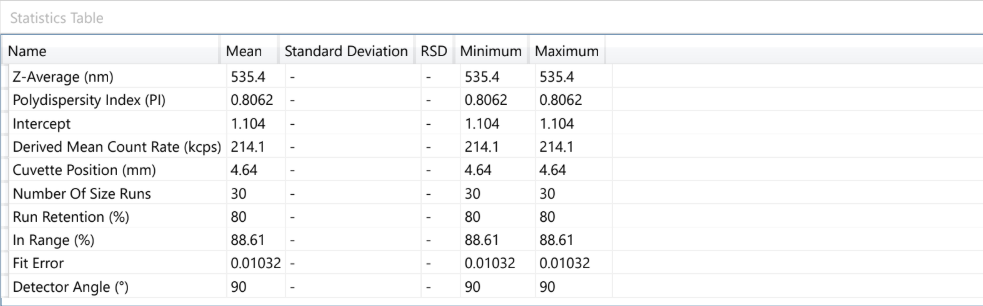
Dynamic Light Scattering (DLS) is employed to evaluate the particle size distribution and polydispersity of liposomal formulations, providing essential information on colloidal stability, dispersion uniformity, and potential bioavailability.
For example – Lipoza®-Curcumin
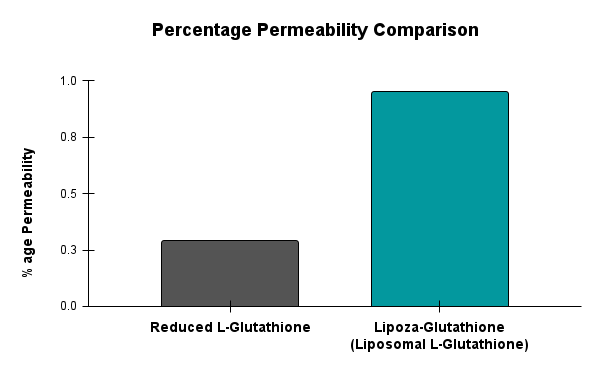
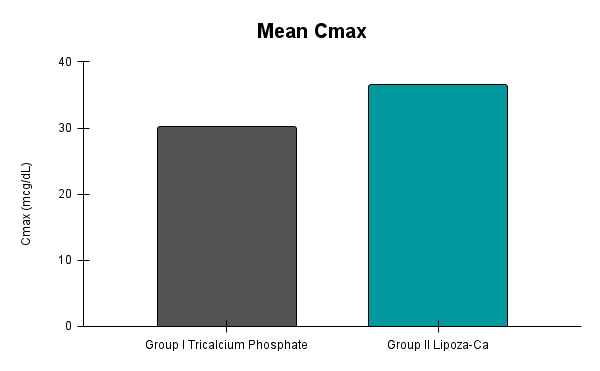
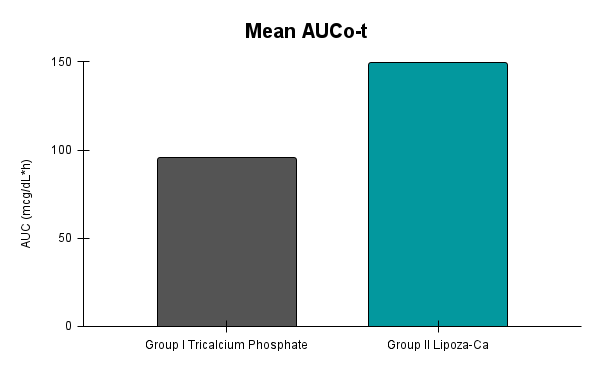
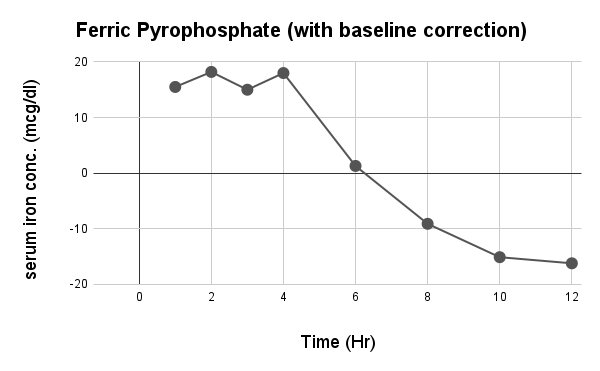
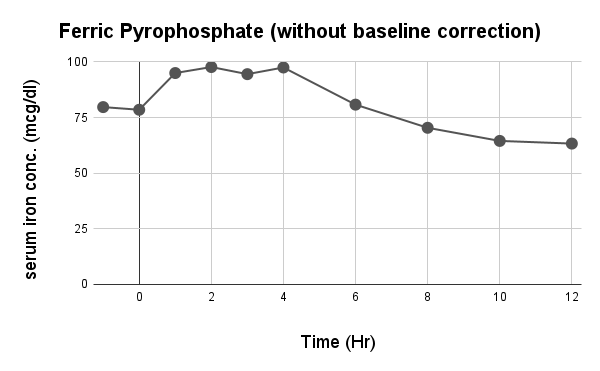
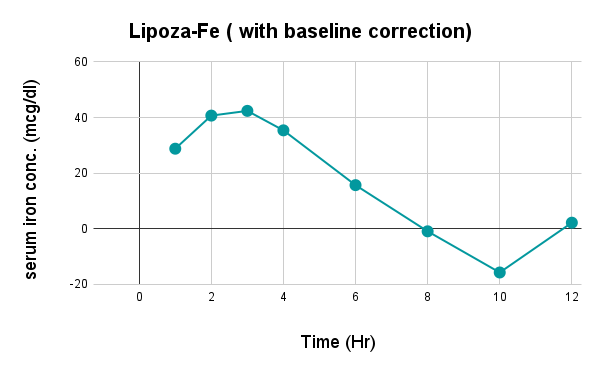
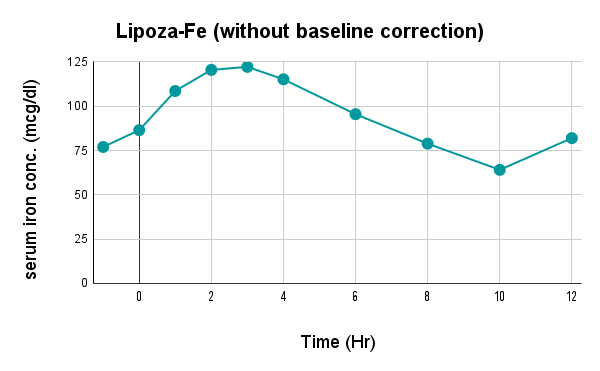
Caco-2 studies utilize human intestinal epithelial cell monolayers to evaluate the permeability and predicted absorption of test substances across the gut barrier.
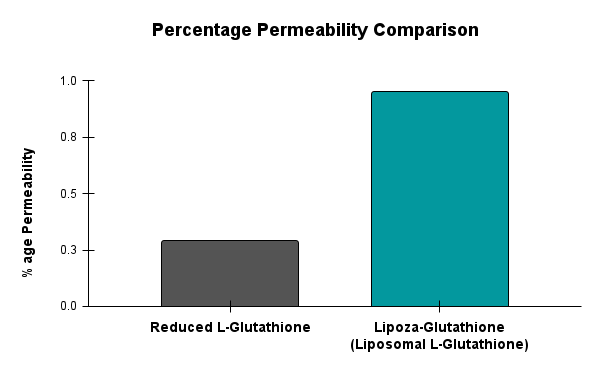
For example – Lipoza-Glutathione
Demonstrated superior permeability, showing 0.95% transport compared to 0.29% for Reduced L-Glutathione—an approximate 3.28-fold increase. This enhanced permeability, despite lower glutathione content, indicates improved intestinal absorption and supports its potential as a more bioavailable form of glutathione supplementation.
Fill out the form below and we'll get back to you soon
* indicates required question