

At OZABIO®, we specialize in the development and manufacturing of premium nutraceutical ingredients designed to address critical healthcare and nutritional challenges. Committed to sustainability, we incorporate eco-friendly technologies throughout our production processes to support a healthier planet and population.
Driven by innovative research and development and a customer-centric philosophy, we strive to deliver advanced solutions that enhance global health outcomes.
As a global leader in high-quality nutritional ingredients, OZABIO® is at the forefront of liposomal innovation. Our state-of-the-art technology enhances the stability and bioavailability of essential nutrients, revolutionizing the way they are absorbed and utilized by the body for maximum health benefits.
With a steadfast dedication to quality, innovation, and customer satisfaction, OZABIO®, is shaping the future of nutritional science—one breakthrough at a time.
To be a global leader in advanced nutraceutical innovation, driving transformative health and well-being with validated, science-backed, high-performance ingredients designed to empower partners and elevate wellness worldwide.
To develop and deliver premium-quality nutraceutical ingredients using cutting-edge technologies, sustainable practices, and rigorous scientific validation—empowering our partners to create effective health solutions for a better tomorrow.






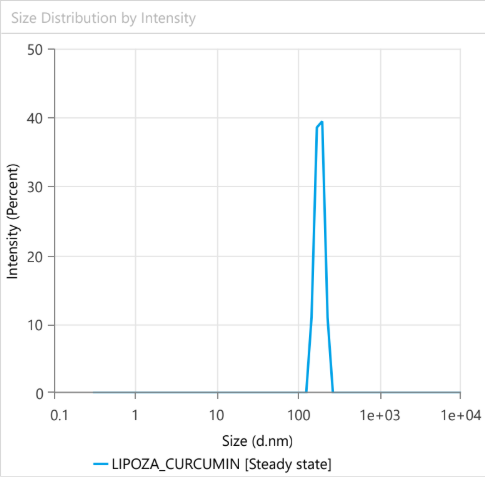
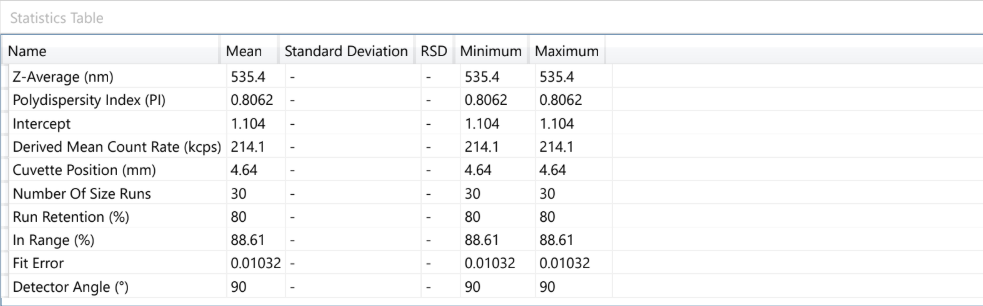
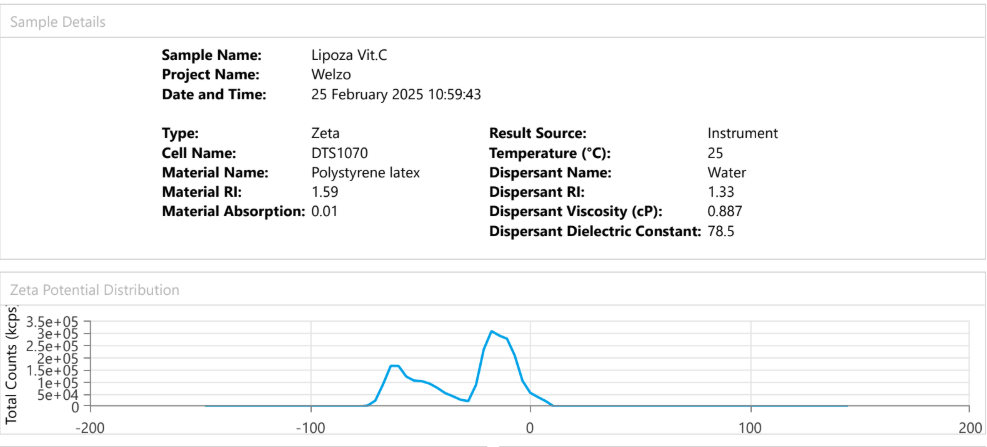
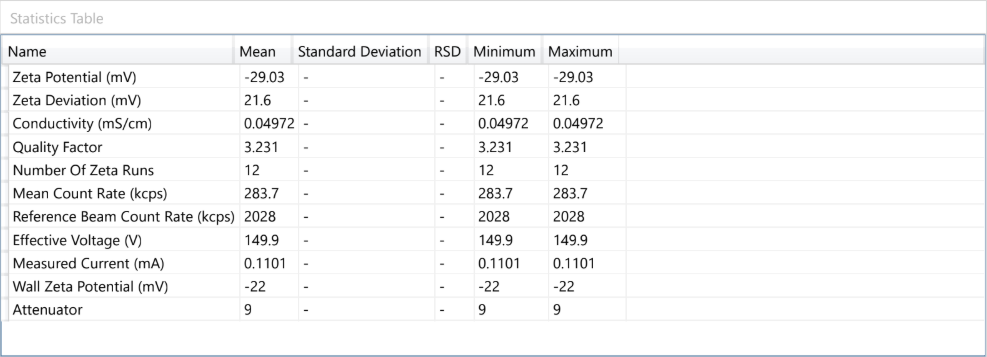
Dynamic Light Scattering (DLS) is employed to evaluate the particle size distribution and polydispersity of liposomal formulations, providing essential information on colloidal stability, dispersion uniformity, and potential bioavailability.
For example – Lipoza®-Curcumin
Caco-2 studies utilize human intestinal epithelial cell monolayers to evaluate the permeability and predicted absorption of test substances across the gut barrier.
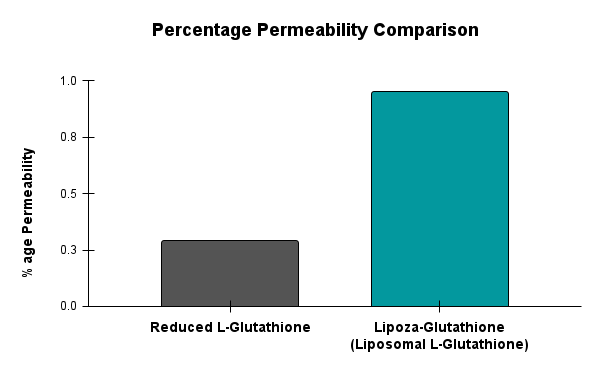
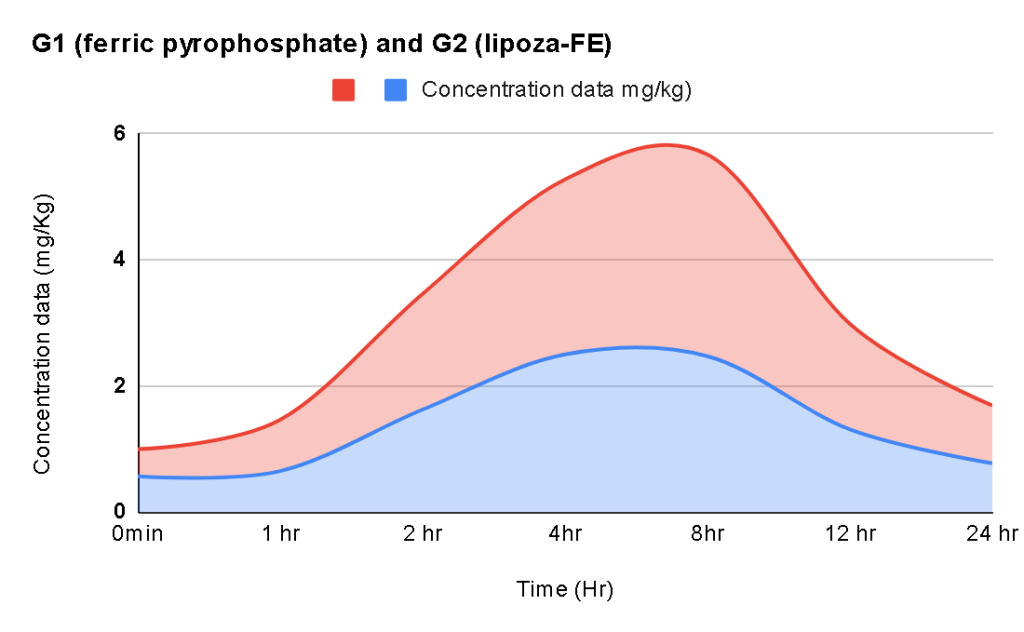
For example – Lipoza-Glutathione
Demonstrated superior permeability, showing 0.95% transport compared to 0.29% for Reduced L-Glutathione—an approximate 3.28-fold increase. This enhanced permeability, despite lower glutathione content, indicates improved intestinal absorption and supports its potential as a more bioavailable form of glutathione supplementation.
Fill out the form below and we'll get back to you soon
* indicates required question